FDA Approves Dupixent for Chronic Spontaneous Urticaria in Young Children
 via HealthDay
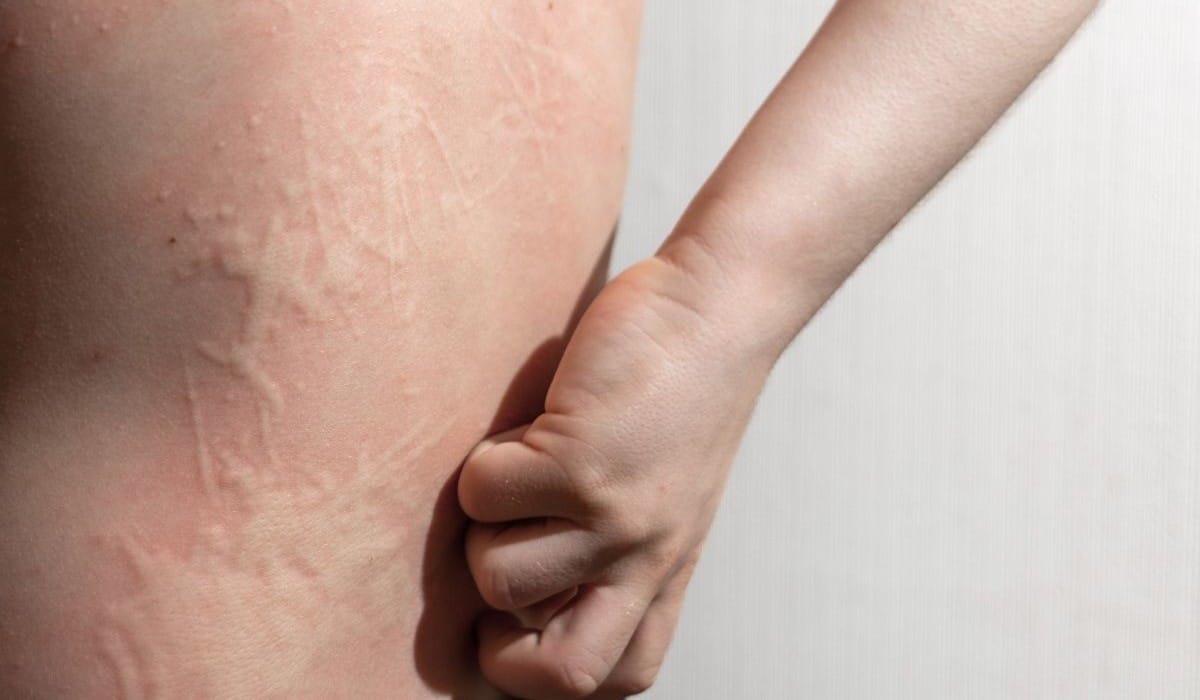
via HealthDayMONDAY, May 4, 2026 -- The U.S. Food and Drug Administration has approved Dupixent (dupilumab) for the treatment of children (aged 2 to 11 years) with chronic spontaneous urticaria (CSU).
This approval expands the previous approval for adults and adolescents ages 12 years and older with CSU who remain symptomatic despite histamine-1 antihistamine treatment. The fully human monoclonal antibody is administered as a subcutaneous injection.
Data from the LIBERTY-CUPID clinical study program informed the approval, which included efficacy and safety data from two phase 3 studies (study A and study C) in adults and adolescents aged 12 years and older with CSU, as well as pharmacokinetics data from the CUPIDKids phase 3 study in children aged 2 to 11 years with CSU. Results from study A and study C showed that Dupixent significantly reduced itch severity and urticaria activity (a composite of itch and hives) versus placebo at week 24 and also increased the likelihood of well-controlled disease or complete response versus placebo. The safety of Dupixent was supported by data from pediatric patients in other indications.
"Dupixent is the first biologic approved for patients as young as 2 years of age, offering a targeted approach that inhibits IL-4 and IL-13 signaling, two key and central drivers of the type 2 inflammation that contributes to this disease," Alyssa Johnsen, M.D., Ph.D., from Sanofi, the manufacturer of Dupixent, said in a statement. "Until now, these patients had to rely on limited treatment options that didn't address potential critical mediators of chronic spontaneous urticaria."
Disclaimer: Statistical data in medical articles provide general trends and do not pertain to individuals. Individual factors can vary greatly. Always seek personalized medical advice for individual healthcare decisions.
Source: HealthDay
Posted : 2026-05-05 09:26
Read more

- Oral Small-Molecule GLP-1 Drugs Penetrate Deep Into the Brain to Suppress Cravings
- Sunscreen Confusion Puts More Americans At Risk For Melanoma
- Weight Loss Surgeries Fall More Than 20% As Patients Turn To GLP-1 Meds, Experts Say
- Coya Therapeutics Has Been Granted U.S. FDA Fast Track Designation for COYA 302 for the Treatment of Amyotrophic Lateral Sclerosis (ALS)
- Opioid OD Survivors Have Triple Rate Of Repeat Overdoses Than Previously Estimated
- Partial Meniscectomy for Degenerative Tear Shows No Benefit at 10 Years
Disclaimer
Every effort has been made to ensure that the information provided by Drugslib.com is accurate, up-to-date, and complete, but no guarantee is made to that effect. Drug information contained herein may be time sensitive. Drugslib.com information has been compiled for use by healthcare practitioners and consumers in the United States and therefore Drugslib.com does not warrant that uses outside of the United States are appropriate, unless specifically indicated otherwise. Drugslib.com's drug information does not endorse drugs, diagnose patients or recommend therapy. Drugslib.com's drug information is an informational resource designed to assist licensed healthcare practitioners in caring for their patients and/or to serve consumers viewing this service as a supplement to, and not a substitute for, the expertise, skill, knowledge and judgment of healthcare practitioners.
The absence of a warning for a given drug or drug combination in no way should be construed to indicate that the drug or drug combination is safe, effective or appropriate for any given patient. Drugslib.com does not assume any responsibility for any aspect of healthcare administered with the aid of information Drugslib.com provides. The information contained herein is not intended to cover all possible uses, directions, precautions, warnings, drug interactions, allergic reactions, or adverse effects. If you have questions about the drugs you are taking, check with your doctor, nurse or pharmacist.
Popular Keywords
- metformin obat apa
- alahan panjang
- glimepiride obat apa
- takikardia adalah
- erau ernie
- pradiabetes
- besar88
- atrofi adalah
- kutu anjing
- trakeostomi
- mayzent pi
- enbrel auto injector not working
- enbrel interactions
- lenvima life expectancy
- leqvio pi
- what is lenvima
- lenvima pi
- empagliflozin-linagliptin
- encourage foundation for enbrel
- qulipta drug interactions
