Hair Growth Product Recalled Over Child Poisoning Risk
 via HealthDay
via HealthDayTHURSDAY, April 9, 2026 — A hair growth product is being recalled because it could pose a serious risk to kids.
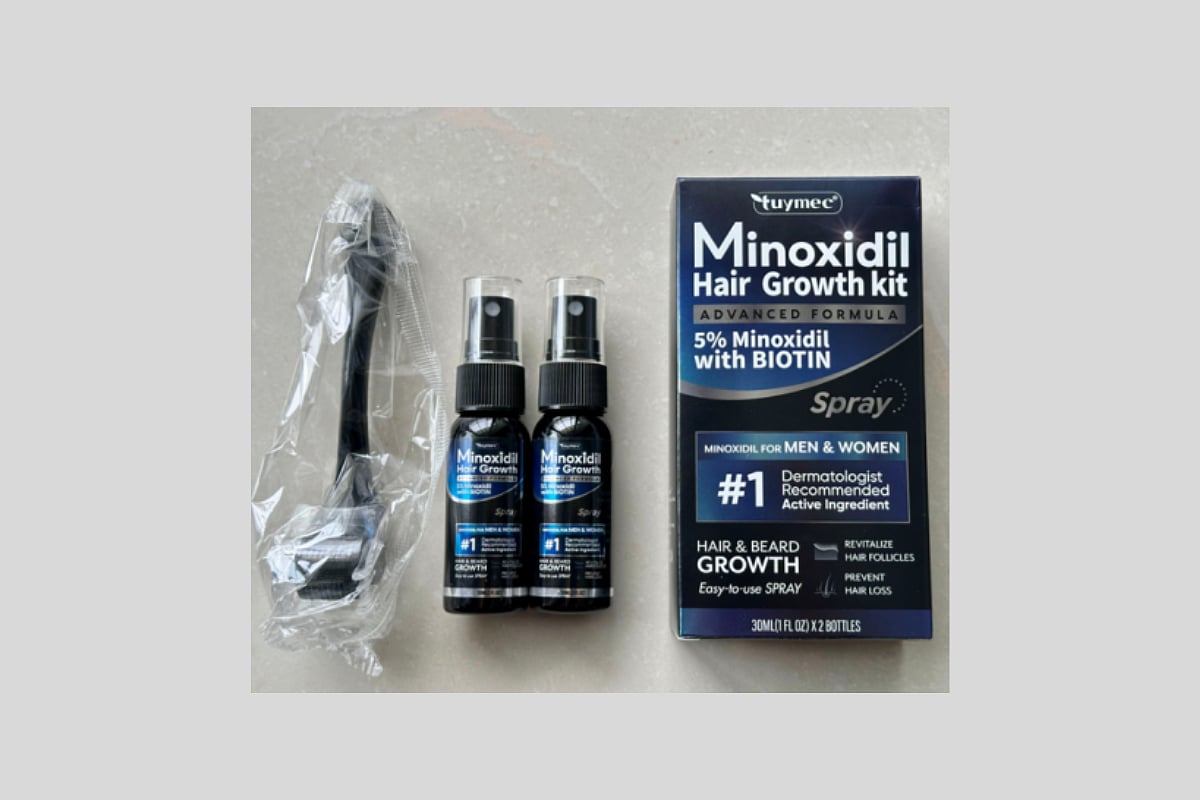
The recall affects Tuymec Minoxidil Hair Growth Kits, which contain a 5% minoxidil spray with biotin, the U.S. Consumer Product Safety Commission (CPSC) said.
CPSC said the bottles are not child-resistant, which could increase the risk of injury or death if a child swallows the liquid inside.
Minoxidil is a medication used to treat hair loss, but it can be harmful if ingested.
As required by the Poison Prevention Packaging Act, products like this must be sold in child-resistant packaging.
The current packaging does not meet those safety standards, CPSC said.
The recall includes about 6,200 units sold on Amazon between June and August of last year for about $24.
The kits include two black spray bottles labeled with the following:
They were made in China and sold by DrHealBeauty of Japan, the CPSC said.
Anyone with the product should stow it out of children’s reach right away.
Contact DrHealBeauty to request a free replacement bottle with a child-resistant cap and then transfer the liquid into the new container.
Customers can reach out via email at [email protected], or contact the company online at drhealbeauty.com and click “Recall Details” at the top of the page for more information.
No injuries have been reported.
Sources
Disclaimer: Statistical data in medical articles provide general trends and do not pertain to individuals. Individual factors can vary greatly. Always seek personalized medical advice for individual healthcare decisions.
Source: HealthDay
Posted : 2026-04-10 01:59
Read more

- Study Shows BMI Often Gets Your Weight Category Wrong
- New White House Budget Plan Would Reduce HHS Funding by Billions
- Wegovy Maker Launches Lower-Cost Subscription Plans
- The White House Delays CDC Pick
- Endotracheal Poractant Alfa Not Effective in Critical Bronchiolitis
- Addition of Atezolizumab to mFOLFOX6 Ups DFS in Stage III dMMR Colon Cancer
Disclaimer
Every effort has been made to ensure that the information provided by Drugslib.com is accurate, up-to-date, and complete, but no guarantee is made to that effect. Drug information contained herein may be time sensitive. Drugslib.com information has been compiled for use by healthcare practitioners and consumers in the United States and therefore Drugslib.com does not warrant that uses outside of the United States are appropriate, unless specifically indicated otherwise. Drugslib.com's drug information does not endorse drugs, diagnose patients or recommend therapy. Drugslib.com's drug information is an informational resource designed to assist licensed healthcare practitioners in caring for their patients and/or to serve consumers viewing this service as a supplement to, and not a substitute for, the expertise, skill, knowledge and judgment of healthcare practitioners.
The absence of a warning for a given drug or drug combination in no way should be construed to indicate that the drug or drug combination is safe, effective or appropriate for any given patient. Drugslib.com does not assume any responsibility for any aspect of healthcare administered with the aid of information Drugslib.com provides. The information contained herein is not intended to cover all possible uses, directions, precautions, warnings, drug interactions, allergic reactions, or adverse effects. If you have questions about the drugs you are taking, check with your doctor, nurse or pharmacist.
Popular Keywords
- metformin obat apa
- alahan panjang
- glimepiride obat apa
- takikardia adalah
- erau ernie
- pradiabetes
- besar88
- atrofi adalah
- kutu anjing
- trakeostomi
- mayzent pi
- enbrel auto injector not working
- enbrel interactions
- lenvima life expectancy
- leqvio pi
- what is lenvima
- lenvima pi
- empagliflozin-linagliptin
- encourage foundation for enbrel
- qulipta drug interactions
